Abstract
Natural lipopeptide antibiotic tripropeptin C (TPPC) revitalizes and synergistically potentiates the activities of the class of β-lactam antibiotics against methicillin-resistant Staphylococcus aureus (MRSA) but not against methicillin-sensitive S. aureus in vitro; however, the mode of action remains unclear. In the course of the study to reveal its mode of action, we found that TPPC inhibited the β-lactamase production induced by cefotiam. This prompted us to focus on the β-lactam-inducible β-lactam-resistant genes blaZ (β-lactamase) and mecA (foreign penicillin-binding protein), as they are mutually regulated by the blaZ/I/R1 and mecA/I/R1 systems. Quantitative reverse-transcription polymerase chain reaction analysis revealed that TPPC reversed β-lactam resistance by reducing the expression of the genes blaZ and mecA, when treated alone or in combination with β-lactam antibiotics. In a mouse/MRSA septicemia model, subcutaneous injection of a combination of TPPC and ceftizoxime demonstrated synergistic therapeutic efficacy compared with each drug alone. These observations strongly suggested that reverse β-lactam resistance by TPPC may be a potentially effective new therapeutic strategy to overcome refractory MRSA infections.
Similar content being viewed by others
Introduction
Methicillin-resistant Staphylococcus aureus (MRSA) strains have acquired multidrug resistance and are one of the major etiological agents of both hospital- and community-acquired infections.1, 2, 3, 4 The high-level resistance of MRSA to β-lactams is caused by acquisition of the foreign penicillin-binding protein (PBP2A; encoded by mecA), to which β-lactams have remarkably low binding affinity.5, 6, 7 In addition to the mecA gene, ~90% of MRSA strains have also acquired a β-lactamase (penicillinase, BlaZ, encoded by blaZ) for penicillin resistance.8, 9 Recently, some MRSA strains have become resistant even to the 'last resort' antibiotic vancomycin (VAN).10, 11, 12 Therefore, new effective drugs and strategies to overcome MRSA are urgently needed.
Tripropeptin C (TPPC) (Figure 1) is a natural, calcium-ion-dependent lipopeptide, which showed potent ant-imicrobial activity against MRSA both in vitro and in a mouse/MRSA ATCC33591-septicemia model when administered intravenously.13, 14, 15 TPPC inhibits peptidoglycan biosynthesis through a different mechanism than that of other drugs targeting peptidoglycan biosynthesis, such as VAN, β-lactams and bacitracin, and shows no cross-resistance with these drugs.15 Recently, we discovered an additional and unique effect of TPPC on revitalizing and synergistically potentiating the activity of the class of β-lactams against MRSA strains.16, 17 Interestingly, the synergy was observed against MRSA but not against methicillin-sensitive S. aureus (MSSA). We previously reported the synergistic anti-MRSA activity of the TPPC/β-lactam combination based on the fractional inhibitory concentration index and time-kill kinetics against MRSA strains, and the mechanism responsible for this effect was suggested to be distinct from its own anti-MRSA activity based on a study using an inactive TPPC semisynthetic derivative.17 However, the mode of action mediating this interesting effect remains unclear.
The purpose of this study was to evaluate whether the synergy of TPPC/β-lactam treatment could also be demonstrated in an in vivo model of mouse MRSA septicemia and to determine the mechanism by which TPPC revitalizes the activity of β-lactams against MRSA. In this study, the in vivo efficacy of the TPPC/β-lactam combination, the inhibition of TPPC on the enzymatic activity and induction of the β-lactamase, BlaZ, and the transcription of β-lactam-inducible drug-resistant genes, both with TPPC alone and its combination with β-lactams, were evaluated. Clinically important anti-MRSA drugs, such as VAN and arbekacin (ABK), were also evaluated for comparison to provide further insight into the mode of revitalization of β-lactams by TPPC. The effects of TPPC/β-lactams against constructed MSSA strains harboring the plasmid containing the mecA gene were examined to reveal the role of this gene or its product.
Materials and methods
Plasmids, bacterial strains and growth conditions
The plasmids and bacterial strains used in this study are listed in Table 1. The shuttle vector pHY300PLK was purchased from Takara (Shiga, Japan) and the β-lactamase gene was removed for this study. All parental strains are commercially available. S.aureus strains were grown at 37 °C with shaking at 140 r.p.m. in Mueller–Hinton broth (Becton Dickinson and Company, Franklin Lakes, NJ, USA) supplemented with 50 μg ml−1 calcium ion (MHCa broth). Recombinant S.aureusstrains were selected and maintained with 10 μg ml−1 tetracycline.
Test compounds
TPPC was prepared as reported previously with slight modifications.15 ABK was kindly provided by Meiji Seika Pharma (Tokyo, Japan). VAN was purchased from Shionogi (Osaka, Japan). Cefotiam (CTM), sulbactam and clavulanic acid were purchased from Wako Pure Chemical Industries (Osaka, Japan). Ceftizoxime (ZOX) was purchased from Fujisawa Pharmaceutical (Osaka, Japan).
In vivo efficacy of TPPC/ZOX in a model of mouse MRSA (ATCC33591) septicemia
Crl:CD1 (ICR) 4-week-old male mice (Charles River Laboratories Japan, Yokohama, Japan) were inoculated intraperitoneally with a lethal dose of MRSA ATCC33591 (4.8 × 107 colony-forming units per mouse) in 0.5 ml of brain heart infusion broth containing 5% mucin. TPPC, ZOX or a combination of both was dissolved in 10% dimethyl sulfoxide saline, and administered subcutaneously at 50, 25 and/or 12.5 mg kg−1. TPPC was administered at 2 h and ZOX was administered two times at 2 and 6 h after the MRSA challenge. Mortality was recorded once daily for 9 days.
Evaluations of the MICs and FICs
MICs and fractional inhibitory concentration indices (FICIs) were evaluated as reported previously.17 FICs were determined using the standard checkerboard methodology18 in MHCa broth and were calculated using the following formulae: FIC of agent A=(MIC of agent A in combination)/(MIC of agent A alone); FIC of agent B=(MIC of agent B in combination)/(MIC of agent B alone); and FIC index (FICI)=(FIC of agent A)+(FIC of agent B).
The combined effects were defined as follows: synergistic: FICI⩽0.5; additive: 0.5
Evaluation of the inhibitory activity of TPPC on β-lactamase enzymatic activity and induction
Nitrocefin was used for evaluating β-lactamase enzymatic activity by measuring the absorbance at 490 nm (iMark Microplate Reader; Bio-Rad, Hercules, CA, USA). Nitrocefin is a chromogenic substrate for β-lactamase, which rapidly turns from yellow to red following β-lactamase degradation.19
The inhibition of TPPC on β-lactamase enzymatic activity was measured as follows. MRSA strain ATCC33591 was grown in MHCa broth at 37 °C with shaking until mid-log phase (OD600 0.5). To this cultured broth, 1 μg ml−1 of CTM (MIC: 512 μg ml−1) was added to induce β-lactamase, if needed. All of the concentrations of CTM tested (0.25–8 μg ml−1) strongly induced the β-lactamase production to a similar extent (data not shown). The broth was cultured for a further 2 h at 37 °C with shaking, and then plated at 200 μl per well in a 96-well microtiter plate. TPPC was added to the wells at final concentrations of 100, 10, 1 and 0.1 μg ml−1, followed by the addition of 100 μg ml−1 of nitrocefin. After 30 min incubation at 37 °C, the absorbance at 490 nm was measured. Sulbactam and clavulanic acid were used as positive controls.
The inhibition of TPPC on the induction of β-lactamase by adding the β-lactam antibiotic CTM was examined as follows. MRSA ATCC33591 strain was cultivated in MHCa broth at 37 °C with shaking until mid-log phase (OD600 0.5). To cultured broths, 1 μg ml−1 of CTM was added to induce β-lactamase, and TPPC, VAN or ABK was also added, if needed, followed by further culture at 37 °C for 1 h with shaking. Then, the β-lactamase activity of each broth was measured as written above.
DNA manipulation
Polymerase chain reaction (PCR) was performed with PrimeSTAR HS DNA polymerase (Takara). Primers used in this study are listed in Supplementary Table S1. Restriction enzymes were used as recommended by the manufacturer (FastDigest; Thermo Fisher Scientific, Waltham, MA, USA). DNA bands were purified with a Wizard SV Gel and a PCR Clean-Up System (Promega, Madison, WI, USA). Total DNA from the MRSA strain N315 was isolated from a bacterial culture using a Genomic DNA Extraction Mini Kit (RBC Bioscience, Taipei, Taiwan) according to the manufacturer’s instructions using lysostaphin (0.5 mg ml−1) in the lysis step. Plasmid DNA was isolated from Escherichia coli cultures using the QuickLyse Miniprep Kit (Qiagen, Hilden, Germany). For plasmid DNA isolation from S. aureus cultures, cell pellets were treated with a bead beader before using the QuickLyse Miniprep Kit (Qiagen). Vector arms and insert ligation were performed with a DNA Ligation Kit Mighty Mix (Takara).
Total RNA purification and quantitative PCR
Total cellular RNA was extracted with zirconia beads (1 μm, TZ-B125m; Tosoh, Tokyo, Japan) and Trizol (Thermo Fisher Scientific) from mid-exponential phase cells of S. aureus (OD600 0.5) grown in 7 ml of MHCa broth, after 10 min exposure with or without drugs. Crude total RNA samples were treated with DNaseI (Takara) to remove any genomic DNA contamination in the presence of recombinant RNase inhibitor (Takara). Transcriptional modulation induced by CTM, ZOX, TPPC or their combinations were investigated by quantitative reverse-transcription PCR using the One Step SYBR PrimeScript Reverse-Transcription-PCR Kit II (Prefect Real Time; Takara) according to the manufacturer’s instructions. Experiments were performed on a Thermal Cycler Dice Real Time System (model TP800; Takara). The newly designed, or previously published,20, 21, 22 primers used in this study are listed in Supplementary Table S1. The thermal conditions were as follows: 5 min at 42 °C and 10 s at 95 °C, followed by 50 cycles at 98 °C for 5 s and 58 °C for 30 s. To verify the specificity of the PCR amplification products, melting-curve analyses were performed. The housekeeping gene gyrA was amplified for normalization. Gene markers blaZ and mecA were indicative of the β-lactam-resistant response, vraS and pbpB were indicative of cell wall stress and vraD was indicative of membrane stress.22, 23
Construction of mecA-overexpressing MSSA
Recombinant plasmids were constructed in E. coli DH5α (Takara), and then electroporated into the MSSA strain ATCC29213 using the Gene Pulser II Electroporation System (Bio-Rad). The plasmid inserts were confirmed by restriction analysis, PCR and DNA sequencing (FASMAC, Kanagawa, Japan).
To clone the mecA gene, a fragment containing mecA and its native promoter was amplified by PCR with mecA primers (Supplementary Table S1) and extracted total DNA from the MRSA strain N315 was used as the template. The PCR product was then cloned into pSTBlue-1 (pSTBlue-1 Perfect Blunt Cloning Kit; Novagen, Darmstadt, Germany). This recombinant plasmid and pHY300PLK(ΔBla)24 were digested with BamHI and HindIII, and the obtained mecA-containing DNA fragment and vector arm were ligated. The resulting recombinant plasmid (pHY300-pmecA) was confirmed by PCR and sequencing, and then electroporated into the MSSA strain ATCC29213.
Ethics statement
All animal experiments were approved by the Institutional Committee for Animal Experiments at the Institute of Microbial Chemistry (BIKAKEN, Tokyo, Japan) and were performed in accordance with the relevant guidelines and regulations to minimize animal suffering.
Results
MICs and FICIs against MRSA ATCC33591
The MICs against the MRSA strain ATCC33591 of CTM, ZOX, TPPC, VAN and ABK were 512, 1024, 1, 1 and 2 μg ml−1, respectively, as determined by the broth dilution assay. The combination effects of CTM or ZOX with TPPC, ABK or VAN against MRSA strain ATCC33591 are shown in Figures 2a and b. When ZOX or CTM was combined with TPPC, synergistic anti-MRSA activities were observed (minimum FICI=0.26 and 0.38, respectively). Meanwhile, VAN and ABK demonstrated only additives, and their minimum FICIs were 0.63 and 0.75 when combined with CTM and 1.0 and 1.0 when combined with ZOX, respectively.
In vitro combination effects against methicillin-resistant Staphylococcus aureus (MRSA) ATCC33591, and against methicillin-sensitive S. aureus (MSSA) ATCC29213 and its mutant derivatives. Anti-MRSA activity of ceftizoxime and cefotiam was revitalized by tripropeptin C (TPPC) but not by vancomycin or arbekacin. The combination effects of TPPC (black diamonds), vancomycin (open circles) and arbekacin (open triangles) with ceftizoxime (a) or cefotiam (b) against the MRSA strain ATCC33591 in checkerboard analysis are shown. (c) The combination effects of TPPC and cefotiam against the MSSA strain ATCC29213 (open triangles), ATCC29213-vector control (open circles) and ATCC29213-mecA (black triangles) are shown. The fractional inhibitory concentration index (FIC) of each antibiotic combination pair is shown, and a total FIC<0.5 (indicated by a diagonal dotted line) indicates a synergistic combination.
Combination effect of TPPC/ZOX in a mouse MRSA (ATCC33591) septicemia model
The 50% effective dose for subcutaneous injection of TPPC or ZOX alone were 50 and >50 mg kg−1 in this animal model (data not shown). As, among β-lactams, ZOX exhibited the highest synergism in vitro against MRSA ATCC33591 when combined with TPPC,17 ZOX was used in this animal model. Interestingly, despite the finding that TPPC alone (12.5 mg kg−1) exhibited no in vivo anti-MRSA activity, the combined administration of TPPC with sub-50% effective dose doses of ZOX remarkably enhanced the efficacies in a dose-dependent manner, as shown in Figure 3. All MRSA-infected mice survived after the combination administration of 12.5 mg kg−1 of TPPC with 25 mg kg−1 of ZOX. The combination 50% effective dose values of TPPC and ZOX were 12.5 and 13.5 mg kg−1, respectively. This established that the TPPC/β-lactam combination treatment exhibits synergistic anti-MRSA activity both in vitro and in vivo, which prompted us to investigate its mode of action.
Therapeutic efficacy of ceftizoxime (ZOX) and tripropeptin C (TPPC) alone or in combination in a mouse methicillin-resistant Staphylococcus aureus (MRSA) septicemia model. (a) Non-treated control (open squares); ZOX, 12.5 mg kg−1 (solid diamonds); ZOX, 25 mg kg−1 (solid triangles). (b) Non-treated control (open squares); TPPC, 12.5 mg kg−1 (solid diamonds); TPPC, 12.5 mg kg−1+ZOX 25 mg kg−1 (open triangles); TPPC, 12.5 mg kg−1+ZOX, 12.5 mg kg−1 (open circles).
Inhibition of TPPC on β-lactamase activity
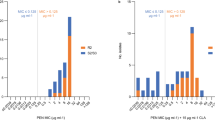
Positive controls clavulanic acid and sulbactam inhibited β-lactamase activity at half-maximal inhibitory concentration of 0.37 and 5.7 μg ml−1, respectively, as shown in Figure 4a. However, TPPC did not inhibit BlaZ β-lactamase activity even at 100 μg ml−1, which was 100 × its MIC.
Inhibition of tripropeptin C (TPPC) and other antibacterials on β-lactamase activity and production in the methicillin-resistant Staphylococcus aureus (MRSA) strain ATCC33591. (a) Effect of TPPC and other inhibitors on β-lactamase activity. TPPC (solid diamonds), clavulanic acid (open triangles) and sulbactam (open circles). Values are expressed as the mean±s.d. for three replicates. (b) Inhibition of TPPC, vancomycin (VAN) and arbekacin (ABK) on cefotiam (CTM)-inducible β-lactamase production. X axis indicates the concentration (μg ml−1) of treated drugs. TPPC (solid diamonds), VAN (open circles) and ABK (open triangles). Values are expressed as the mean±s.d. for three replicates of one of two independent experiments.
Effect of TPPC on β-lactam-inducible β-lactamase production
CTM-induced β-lactamase production was not reduced by VAN or ABK even at concentrations around their MICs, as shown in Figure 4b. By contrast, TPPC reduced CTM-induced β-lactamase production with an half-maximal inhibitory concentration of 0.06 × the MIC, as shown in Figure 4b. Unlike VAN and ABK, TPPC inhibited β-lactam-inducible β-lactamase expression. As the expression of the mecA and blaZ genes is mutually regulated by the mecA/I/R1 and blaZ/I/R1 systems,21 our findings indicated the possibility that the expression of not only blaZ but also mecA may be inhibited by TPPC.
Effect of TPPC on the transcription of drug-response genes
To clarify the inhibition of TPPC on the expression of β-lactam-inducible resistance genes, together with the major two-component regulatory systems involved in membrane and cell wall stress, the transcriptions of gyrA (control), blaZ, blaI, blaR1, mecA, mecI,mecR1, vraS, vraD and pbpB were examined. MRSA strain ATCC33591 was treated with 1/1024 × MIC of CTM, 1/128 × MIC of ZOX and/or 1/2 × MIC of TPPC and the results are shown in Figure 5a and Supplementary Figures S1 and S2. As expected, CTM treatment greatly enhanced the expressions of mecA (3.8-fold) and blaZ (35.4-fold) compared with the non-treated group, whereas changes in expressions of vraS (1.0-fold), pbpB (1.1-fold) and vraD (1.1-fold) were not observed. TPPC alone reduced the expressions of β-lactam-inducible resistance genes mecA (0.2-fold) and blaZ (0.5-fold) and positively induced the expressions of vraS (15.3-fold), vraD (2.0-fold) and pbpB (2.8-fold), which might reflect cell wall biosynthesis inhibition and membrane interactions with TPPC. Even under CTM-induced conditions, the addition of TPPC reduced the expressions of β-lactam-inducible resistance genes blaZ (1.9-fold) and mecA (0.5-fold) to lower than those of CTM alone and near to the levels observed in the absence of CTM. Under these conditions, TPPC enhanced the expressions of vraS (14.4-fold), pbpB (2.6-fold) and vraD (2.0-fold), which were comparable to those of TPPC alone, indicating that TPPC still affects both cell wall biosynthesis and membrane interactions. The combination effects of TPPC with ZOX on the transcriptions of the genes of interest are shown in Supplementary Figure S2, and their results were similar to those of TPPC with CTM.
Effect of tripropeptin C (TPPC) and β-lactams on the transcription of drug-response genes. (a) Effects of TPPC, cefotiam (CTM) and ceftizoxime (ZOX) on the expression of the blaZ and mecA genes in methicillin-resistant Staphylococcus aureus (MRSA) strain ATCC33591. Values are expressed as the mean±s.d. for three replicates of one of three independent experiments. (b) Effects of TPPC and CTM on transcription levels of drug-response genes in methicillin-sensitive S. aureus (MSSA) strain ATCC29213-pmecA. Values are expressed as the mean±s.d. for three replicates of one of two independent experiments.
The effect of VAN on transcription of drug-response genes was also evaluated for comparison (Supplementary Figure S1). VAN enhanced vraS expression (3.6-fold) but did not reduce the expression of β-lactam resistance genes blaZ (1.4-fold) and mecA (1.8-fold). Following VAN/CTM cotreatment, the expressions of blaZ (20.7-fold) and mecA (3.5-fold) were much higher than VAN alone, which was expected as VAN exhibited neither synergistic anti-MRSA activity nor inhibition of the induction of β-lactamase when treated with β-lactams.
Combination effects of β-lactam and TPPC against a mecA-expressing MSSA strain
Since the β-lactam revitalizing activity of TPPC was observed selectively against MRSA but not against MSSA, we then focused on the role of the resistance determinant gene, mecA, to investigate further the difference between MSSA and MRSA. We constructed a mecA-expressing MSSA strain (ATCC29213-pmecA) and evaluated the susceptibility and combinatorial effect of CTM and TPPC against MSSA strain ATCC29213-pmecA transformants (n=5) and the vector-only control strain, ATCC29213-vector (n=3). The MICs of TPPC and CTM against ATCC29213 and its mutant derivatives are summarized in Supplementary Table S3. The mecA-overexpressing mutants acquired resistance against CTM and its MIC was 512 μg ml−1. Meanwhile, ATCC29213-vector retained sensitivity to CTM (MIC: 1 μg ml−1), similar to that of the parent strain. By contrast, the MIC of TPPC against ATCC29213-pmecA (n=5) was 1 μg ml−1, which was the same as its vector control strain ATCC29213-vector. CTM/TPPC combination treatment against ATCC29213-pmecA mutants exhibited synergy and their minimum FICI was 0.28, as shown in Figure 2c. By contrast, no synergy was observed in ATCC29213-vector by CTM/TPPC.
These results prompted us to question whether inhibition of mecA gene expression is affected by the synergistic effects of TPPC/β-lactam cotreatment. And to investigate this, the inhibitions of TPPC/β-lactam cotreatment on the transcriptions of the mecA and blaZ/I/R1 genes in mecA-overexpressing MSSA ATCC29213 were examined. In MSSA ATCC29213-pmecA, blaZ and mecA expression was expected to be β-lactam inducible because of this strain harbors its own blaZ/I/R1 system, and the blaZ (61.4-fold) and mecA (3.3-fold) genes were actually found to be induced by CTM, as shown in Figure 5b. As expected, in response to TPPC treatment alone or in combination with CTM, the expression of the mecA (0.7- and 1.3-fold) and blaZ (0.3- and 0.5-fold) genes was much lower than that of the CTM-treated group, and was comparable to that of the none CTM treatment group, as shown in Figure 5b. These results indicated that TPPC inhibited the transcription of β-lactam-inducible β-lactam resistance genes, leading to reverse β-lactam resistance.
Discussion
In this study, we confirmed the revitalization and synergistic potentiation of β-lactam activity against MRSA by TPPC in an in vivo model of mouse MRSA septicemia. We previously reported that the combination effect of TPPC was observed against only MRSA but not MSSA, and the carboxylic acids in the TPPC molecule are not essential for the synergy with β-lactams, although the existence of carboxylic acids in TPPC is essential to exhibit its potent anti-bacterial activities.25 This indicates that the mode of action of TPPC-reversing β-lactam resistance in MRSA would be different from that of the TPPC-bactericidal effect caused by peptidoglycan biosynthesis inhibition.17 To clarify the mode of revitalization of β-lactams by TPPC, we focused on the β-lactam-inducible resistance factors BlaZ and PBP2A, which are encoded by the blaZ and mecA genes, respectively. Most of MRSA strains have acquired the β-lactamase BlaZ9 that hydrolyses the β-lactam ring and results in deactivation of anti-bacterial properties. We found that TPPC inhibited the BlaZ production induced by cefotiam treatment in the MRSA strain ATCC33591. This indicated that TPPC likely inhibits the expression of the β-lactam-inducible resistance genes blaZ and mecA, as they are mutually regulated by the blaZ/I/R1 and mecA/I/R1 systems.21 A characteristic feature of MRSA is the acquisition of the foreign mecA gene, of which product PBP2A can polymerize peptidoglycan in the presence of β-lactams because of its low affinity to this class of antibiotics. This results in high-level β-lactam resistance. The possibility we proposed in the previous report17 that TPPC inhibited MecA (PBP2A) to potentiate the anti-MRSA activity of β-lactam antibiotics is unlikely, as TPPC exhibited the same MIC against both mecA-overexpressing ATCC29213 and mecA-non-expressing vector control strain. Instead, our results revealed that TPPC reduced the transcription of β-lactam-inducible-β-lactam resistance genes mecA and blaZ to levels comparable to those not induced by β-lactam. However, regardless of the presence of β-lactams, TPPC affected the cell wall and membrane, as deduced by the enhanced expression of vraS, vraD and pbpB (Supplementary Figure S1). The interaction between TPPC and the bacterial membrane was consistent with our previously reported findings that TPPC led to slight membrane depolarization but did not disrupt membrane function.15 These observations demonstrated that TPPC disrupts the drug response specifically caused by β-lactam antibiotics.
In the MSSA strain ATCC29213-pmecA harboring the mecA-expressing plasmid, blaZ,mecA expression was β-lactam-inducible as expected, probably via its own blaZ/I/R1 system. When strain ATCC29213-pmecA was exposed to CTM, TPPC exhibited synergistic anti-MRSA activity and reverse β-lactam resistance, which differed from its effect on MSSA strains17 and the vector control strain ATCC29213-vector (BlaZ positive). This demonstrated that the expression of the β-lactamase blaZ gene alone is insufficient for TPPC to exhibit synergism with β-lactams. By contrast, it was suggested that both mecA expression system and reduction of mecA expression are essential for TPPC-synergistic anti-MRSA activity with β-lactams. In fact, TPPC exhibited reduction of the expression of the β-lactam-inducible resistance gene mecA (and blaZ) in both MRSA ATCC33591 and MSSA ATCC29213-pmecA.
Other researchers have previously reported that blocking the signal pathway or transcription of mecA and blaZ restores β-lactam susceptibility in MRSA.26, 27, 28 Thus, we concluded that TPPC exhibited reverse β-lactam resistance by reducing the expression of inducible-β-lactam resistance genes in mecA-expressing strains, including clinically isolated MRSA strains. On the other hand, how β-lactams potentiate the anti-MRSA activity of TPPC remains to be determined and is under investigation.
Next, we considered how TPPC reduces the expression of both blaZ and mecA. These genes are mutually regulated by the blaZ/I/R1 and mecA/I/R1 systems21 as follows (Figure 6). Binding of the β-lactams to the transmembrane sensor-transducer BlaR1/MecR1 activates their protease activity resulting in inactivation of BlaI/MecI, repressors of blaZ/mecA RNA transcription, and leading to β-lactam-inducible blaZ/mecA expression resulting in β-lactam resistance. Within this process, there are at least three possible TPPC inhibitory steps (Figure 6): (1) TPPC interferes with the sensing of β-lactams by BlaR1/MecR1 or their autocatalytic activations; (2) TPPC inhibits the BlaR1/MecR1 protease activity toward BlaI/MecI or the release of inactivated BlaI/MecI from DNA-binding sites; (3) TPPC selectively inhibits the transcription of blaZ/mecA genes. As demonstrated in Supplementary Figure S2, TPPC treatment reduced the expression of both the mecA/I/R1 and blaZ/I/R1 systems, suggesting that TPPC might block pathways upstream of these signal transductions.
Possible mode of action of reverse β-lactam resistance mediated by tripropeptin C (TPPC). (a) In the absence of β-lactam, DNA-binding proteins of BlaI/MecI bind to the operator regions between the blaZ and blaR1 or mecA and mecR1 genes, which results in repression of the expression of these genes. (b) β-lactam sensing by the transmembrane transducer BlaR1 or MecR1 activates BlaR1 or MecR1 protease activity, which digests and inactivates the BlaI/MecI repressors via blaZ/mecA RNA transcription and leads to blaZ/mecA expression and the resulting β-lactam resistance. (c) Regardless of the presence of β-lactams, TPPC reduced the β-lactam-inducible β-lactam resistance genes. Possible TPPC inhibitory steps are shown in a gray rectangle.
In this study, we revealed that TPPC inhibited the transcription of the β-lactam-inducible resistance genes mecA (PBP2A) and β-lactamase (blaZ) in MRSA, resulting in the reverse β-lactam resistance. However, the target of TPPC remains to be determined, and identification of the target molecule is currently under investigation.
References
Boucher, H. W. et al. Bad bugs, no drugs: no ESKAPE! An update from the infectious diseases society of America. Clin. Infect. Dis. 48, 1–12 (2009).
Klevens, R. M. et al. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. J. Am. Assoc. 298, 1763–1771 (2007).
DeLeo, F. R., Otto, M., Kreiswirth, B. N. & Chambers, H. F. Community-associated methicillin-resistant Staphylococcus aureus. Lancet 375, 1557–1568 (2010).
Otter, J. A. & French, G. L. Molecular epidemiology of community-associated methicillin-resistant Staphylococcus aureus in Europe. Lancet Infect. Dis. 10, 227–239 (2010).
Song, M. D., Wachi, M., Doi, M., Ishino, F. & Matsuhashi, M. Evolution of an inducible penicillin-target protein in methicillin-resistant Staphylococcus aureus by gene fusion. FEBS Lett. 221, 167–171 (1987).
Ubukata, K., Nonoguchi, R., Matsuhashi, M. & Konno, M. Expression and inducibility in Staphylococcus aureus of the mecA gene, which encodes a methicillin-resistant S. aureus-specific penicillin-binding protein. J. Bacteriol. 171, 2882–2885 (1989).
Villegas-Estrada, A., Lee, M., Hesek, D., Vakulenko, S. B. & Mobashery, S. Co-opting the cell wall in fighting methicillin-resistant Staphylococcus aureus: potent inhibition of PBP 2a by two anti-MRSA beta-lactam antibiotics. J. Am. Chem. Soc. 130, 9212–9213 (2008).
Novick, R. P. Staphylococcal penicillinase and the new penicillins. Biochem. J. 83, 229–265 (1962).
Franklin, D. L. Antimicrobial resistance: the example of Staphylococcus aureus. J. Clin. Invest. 111, 1265–1273 (2003).
Hiramatsu, K. et al. Methicillin-resistant Staphylococcus aureus clinical strain with reduced vancomycin susceptibility. J. Antimicrob. Chemother. 40, 135–136 (1997).
Holmes, N. E., Johnson, P. D. R. & Howden, B. P. Relationship between vancomycin-resistant Staphylococcus aureus, vancomycin-intermediate S. aureus, high vancomycin MIC, and outcome in serious S. aureus infections. J. Clin. Microbiol. 50, 2548–2552 (2012).
Moravvej, Z. et al. Update on the global number of vancomycin-resistant Staphylococcus aureus (VRSA) strains. Int. J. Antimicrob. Agents 42, 370–371 (2013).
Hashizume, H. et al. Tripropeptins, novel antimicrobial agents produced by Lysobacter sp. I. Taxonomy, isolation and biological activities. J. Antibiot. 54, 1054–1059 (2001).
Hashizume, H. et al. Tripropeptins, novel antimicrobial agents produced by Lysobacter sp. II. Structure elucidation. J. Antibiot. 57, 52–58 (2004).
Hashizume, H. et al. Tripropeptin C blocks the lipid cycle of cell wall biosynthesis by complex formation with undecaprenyl pyrophosphate.Antimicrob. Agents Chemother. 55, 3821–3828 (2011).
Hashizume, H., Takahashi, Y., Harada, S. & Nomoto, A. Combination effect of natural lipopeptide tripropeptin C and clinically important drugs against MRSA and VRE. in 54th Interscience Conference on Antimicrobial Agents Chemotherapy (Washiongton, DC, abstr., Poster F-255, American Society for Microbiology, 2014).
Hashizume, H., Takahashi, Y., Harada, S. & Nomoto, A. Natural lipopeptide antibiotic tripropeptin C revitalizes and synergistically potentiates the activity of beta-lactam against methicillin-resistant Staphylococcus aureus. J. Antibiot. 68, 373–378 (2015).
Moody, J. Synergism testing: broth microdilution checkerboard and broth macrodilution methods. in Clinical Microbiology Procedures Handbook 2nd edn (eds Garcia, L. S. & Isenberg, H. D.) (ASM Press, Washington, DC, 5.12.1–5.12.23, 2007).
O’Callaghan, C. H., Morris, A., Kirby, S. M. & Shingler, A. H. Novel method for detection of beta-lactamases by using a chromogenic cephalosporin substrate. Antimicrob. Agents Chemother. 1, 283–288 (1972).
Gardete, S., Lencastre, H. & Tomasz, A. A link in transcription between the native pbpB and the acquired mecA gene in a strain of Staphylococcus aureus.Microbiology 152, 2549–2558 (2006).
Hou, Z. et al. Co-blockade of mecR1/blaR1 signal pathway to restore antibiotic susceptibility in clinical isolates of methicillin-resistant Staphylococcus aureus. Arch. Med. Sci. 7, 414–422 (2011).
Pietiainen, M. et al. Transcriptome analysis of the responses of Staphylococcus aureus to antimicrobial peptides and characterization of the roles of vraDE and vraSR in antimicrobial resistance. BMC Genomics 10, 429 (2009).
Kawada-Matsuo, M., Yoshida, Y., Nakamura, N. & Komatsuzawa, H. Role of two-component systems in the resistance of Staphylococcus aureus to antibacterial agents. Virulence 2, 427–430 (2011).
Ishizaki, Y. et al. Inhibition of the first step in synthesis of the mycobacterial cell wall core, catalyzed by the GlcNAc-1-phosphate transferase WecA, by the novel caprazamycin derivative CPZEN-45. J. Biol. Chem. 288, 30309–30319 (2013).
Hirosawa, S., Takahashi, Y., Hashizume, H., Miyake, T. & Akamatsu, Y. Synthesis and antibacterial activity of tripropeptin C derivatives modified at the carboxyl groups. J. Antibiot. 67, 265–268 (2014).
Meng, J. et al. Restoration of oxacillin susceptibility in methicillin-resistant Staphylococcus aureus by blocking the MecR1-mediated signaling pathway. J. Chemother. 18, 360–365 (2006).
Meng, J. et al. Reversion of antibiotic resistance by inhibiting mecA in clinical methicillin-resistant Staphylococci by antisense phosphorothioate oligonucleotide. J. Antibiot. 68, 158–164 (2015).
Qin, W., Panunzio, M. & Biondi, S. Beta-lactam antibiotics renaissance. Antibiotics 3, 193–215 (2014).
Acknowledgements
The authors greatly appreciate the valuable advice from Prof Akio Nomoto and Dr Yoshimasa Ishizaki and the technical support from Shigeko Harada and Akiko Harakawa at the Institute of Microbial Chemistry (BIKAKEN, Japan).
This work was supported by the Japan Society for the Promotion of Science KAKENHI, grant numbers 24710254 and 16K08338.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This article is dedicated to Prof. Hamao Umezawa on the occasion of the 60th anniversary of worldwide marketing of kanamycin.
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Hashizume, H., Takahashi, Y., Masuda, T. et al. In vivo efficacy of β-lactam/tripropeptin C in a mouse septicemia model and the mechanism of reverse β-lactam resistance in methicillin-resistant Staphylococcus aureus mediated by tripropeptin C. J Antibiot 71, 79–85 (2018). https://doi.org/10.1038/ja.2017.88
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2017.88
This article is cited by
-
Precursor-directed biosynthesis and biological activity of tripropeptin Cpip, a new tripropeptin C analog containing pipecolic acid
The Journal of Antibiotics (2024)
-
Subinhibitory Levels of Fluoroquinolones Result in Enrichment of the Membrane Proteome of Staphylococcus aureus
Journal of Ocean University of China (2023)
-
Lipid oligonucleotides as a new strategy for tackling the antibiotic resistance
Scientific Reports (2020)