Abstract
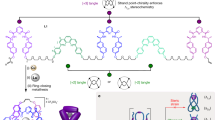
Current strategies for the synthesis of molecular knots focus on twisting, folding and/or threading molecular building blocks. Here we report that Zn(ii) or Fe(ii) ions can be used to weave ligand strands to form a woven 3 × 3 molecular grid. We found that the process requires tetrafluoroborate anions to template the assembly of the interwoven grid by binding within the square cavities formed between the metal-coordinated criss-crossed ligands. The strand ends of the grid can subsequently be joined through within-grid alkene metathesis reactions to form a topologically trivial macrocycle (unknot), a doubly interlocked [2]catenane (Solomon link) and a knot with seven crossings in a 258-atom-long closed loop. This 74 knot topology corresponds to that of an endless knot, which is a basic motif of Celtic interlace, the smallest Chinese knot and one of the eight auspicious symbols of Buddhism and Hinduism. The weaving of molecular strands within a discrete layer by anion-template metal–ion coordination opens the way for the synthesis of other molecular knot topologies and to woven polymer materials.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Crystallographic data have been deposited at the Cambridge Crystallographic Data Centre (http://www.ccdc.cam.ac.uk/) under CCDC number 2022144. These data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within the paper and its Supplementary Information, or are available from the Mendeley data repository at https://doi.org/10.17632/g5b854f68w.1.
References
Ashley, C. The Ashley Book of Knots (Doubleday, 1994).
Fielden, S. D. P., Leigh, D. A. & Woltering, S. L. Molecular knots. Angew. Chem. Int. Ed. 56, 11166–11194 (2017).
Frank-Kamenetskii, M. D., Lukashin, A. V. & Vologodskii, A. V. Statistical mechanics and topology of polymer chains. Nature 258, 398–402 (1975).
Lim, N. C. H. & Jackson, S. E. Molecular knots in biology and chemistry. J. Phys. Condens. Matter 27, 354101 (2015).
Saitta, A. M., Soper, P. D., Wasserman, E. & Klein, M. L. Influence of a knot on the strength of a polymer strand. Nature 399, 46–48 (1999).
Katsonis, N. et al. Knotting a molecular strand can invert macroscopic effects of chirality. Nat. Chem. 12, 939–944 (2020).
Benyettou, F. et al. Potent and selective in vitro and in vivo antiproliferative effects of metal–organic trefoil knots. Chem. Sci. 10, 5884–5892 (2019).
Ayme, J.-F. et al. Strong and selective anion binding within the central cavity of molecular knots and links. J. Am. Chem. Soc. 137, 9812–9815 (2015).
Bilbeisi, R. A. et al. [C–H···anion] interactions mediate the templation and anion binding properties of topologically non-trivial metal–organic structures in aqueous solutions. Chem. Sci. 7, 2524–2532 (2016).
Marcos, V. et al. Allosteric initiation and regulation of catalysis with a molecular knot. Science 352, 1555–1559 (2016).
Gil-Ramírez, G. et al. Tying a molecular overhand knot of single handedness and asymmetric catalysis with the corresponding pseudo-D3-symmetric trefoil knot. J. Am. Chem. Soc. 138, 13159–13162 (2016).
Dietrich-Buchecker, C. O. & Sauvage, J.-P. A synthetic molecular trefoil knot. Angew. Chem. Int. Ed. Engl. 28, 189–192 (1989).
Guo, J., Mayers, P. C., Breault, G. A. & Hunter, C. A. Synthesis of a molecular trefoil knot by folding and closing on an octahedral coordination template. Nat. Chem. 2, 218–222 (2010).
Barran, P. E. et al. Active metal template synthesis of a molecular trefoil knot. Angew. Chem. Int. Ed. 50, 12280–12284 (2011).
Ayme, J.-F. et al. A synthetic molecular pentafoil knot. Nat. Chem. 4, 15–20 (2012).
Ayme, J.-F. et al. Lanthanide template synthesis of a molecular trefoil knot. J. Am. Chem. Soc. 136, 13142–13145 (2014).
Zhang, L. et al. A molecular trefoil knot from a trimeric circular helicate. J. Am. Chem. Soc. 140, 4982–4985 (2018).
Danon, J. J. et al. Braiding a molecular knot with eight crossings. Science 355, 159–162 (2017).
Zhang, L. et al. Stereoselective synthesis of a composite knot with nine crossings. Nat. Chem. 10, 1083–1088 (2018).
Leigh, D. A. et al. Tying different knots in a molecular strand. Nature 584, 562–568 (2020).
Segawa, Y. et al. Topological molecular nanocarbons: all-benzene catenane and trefoil knot. Science 365, 272–276 (2019).
Safarowsky, O., Nieger, M., Fröhlich, R. & Vögtle, F. A molecular knot with twelve amide groups—one-step synthesis, crystal structure, chirality. Angew. Chem. Int. Ed. 39, 1616–1618 (2000).
Feigel, M., Ladberg, R., Engels, S., Herbst-Irmer, R. & Fröhlich, R. A trefoil knot made of amino acids and steroids. Angew. Chem. Int. Ed. 45, 5698–5702 (2006).
Kim, D. H. et al. Coordination-driven self-assembly of a molecular knot comprising sixteen crossings. Angew. Chem. Int. Ed. 57, 5669–5673 (2018).
Leigh, D. A., Lemonnier, J.-F. & Woltering, S. L. Comment on “Coordination-driven self-assembly of a molecular knot comprising sixteen crossings”. Angew. Chem. Int. Ed. 57, 12212–12214 (2018).
Inomata, Y., Sawada, T. & Fujita, M. Metal–peptide torus knots from flexible short peptides. Chem 6, 294–303 (2020).
Ponnuswamy, N., Cougnon, F. B. L., Clough, J. M., Pantoş, G. D. & Sanders, J. K. M. Discovery of an organic trefoil knot. Science 338, 783–785 (2012).
Ponnuswamy, N., Cougnon, F. B. L., Pantoş, G. D. & Sanders, J. K. M. Homochiral and meso figure eight knots and a Solomon link. J. Am. Chem. Soc. 136, 8243–8251 (2014).
Cougnon, F. B. L., Caprice, K., Pupier, M., Bauza, A. & Frontera, A. A strategy to synthesize molecular knots and links using the hydrophobic effect. J. Am. Chem. Soc. 140, 12442–12450 (2018).
Forgan, R. S., Sauvage, J.-P. & Stoddart, J. F. Chemical topology: complex molecular knots, links, and entanglements. Chem. Rev. 111, 5434–5464 (2011).
Ayme, J.-F., Beves, J. E., Campbell, C. J. & Leigh, D. A. Template synthesis of molecular knots. Chem. Soc. Rev. 42, 1700–1712 (2013).
Sauvage, J.-P. From chemical topology to molecular machines (Nobel Lecture). Angew. Chem. Int. Ed. 56, 11080–11093 (2017).
Marenda, M., Orlandini, E. & Micheletti, C. Discovering privileged topologies of molecular knots with self-assembling models. Nat. Commun. 9, 3051 (2018).
Alexander, J. W. & Briggs, G. B. On types of knotted curves. Ann. Math. 28, 562–586 (1926).
Bucka, A. & Stasiak, A. Construction and electrophoretic migration of single-stranded DNA knots and catenanes. Nucleic Acids Res. 30, e24 (2002).
Ruben, M., Rojo, J., Romero‐Salguero, F. J., Uppadine, L. H. & Lehn, J.-M. Grid‐type metal ion architectures: functional metallosupramolecular arrays. Angew. Chem. Int. Ed. 43, 3644–3662 (2004).
Dawe, L. N., Abedin, T. S. M. & Thompson, L. K. Ligand directed self-assembly of polymetallic [n × n] grids: rational routes to large functional molecular subunits? Dalton Trans. 2008, 1661–1675 (2008).
Beves, J. E., Danon, J. J., Leigh, D. A., Lemonnier, J.-F. & Vitorica-Yrezabal, I. J. A Solomon link through an interwoven molecular grid. Angew. Chem. Int. Ed 54, 7555–7559 (2015).
Danon, J. J., Leigh, D. A., Pisano, S., Valero, A. & Vitorica-Yrezabal, I. J. A six-crossing doubly interlocked [2]catenane with twisted rings, and a molecular granny knot. Angew. Chem. Int. Ed. 57, 13833–13837 (2018).
Bianchi, A., Bowman-James, K. & Garcia, E. Supramolecular Chemistry of Anions (Wiley-VCH, 1997).
August, D. P. et al. Self-assembly of a layered 2D molecularly woven fabric. Nature https://doi.org/10.1038/s41586-020-3019-9 (2020).
Garber, S. B., Kingsbury, J. S., Gray, B. L. & Hoveyda, A. H. Efficient and recyclable monomeric and dendritic Ru-based metathesis catalysts. J. Am. Chem. Soc. 122, 8168–8179 (2000).
De Gennes, P. G. Reptation of a polymer chain in the presence of fixed obstacles. J. Chem. Phys. 55, 572–571 (1971).
Kidd, T. J., Leigh, D. A. & Wilson, A. J. Organic ‘magic rings’—the hydrogen bond-directed assembly of catenanes under thermodynamic control. J. Am. Chem. Soc. 121, 1599–1600 (1999).
Chichak, K. S. et al. Molecular Borromean rings. Science 304, 1308–1312 (2004).
Leigh, D. A., Pritchard, R. G. & Stephens, A. J. A Star of David catenane. Nat. Chem. 6, 978–982 (2014).
Hubin, T. J. & Busch, D. H. Template routes to interlocked molecular structures and orderly molecular entanglements. Coord. Chem. Rev. 200–202, 5–52 (2000).
Cockriel, D. L. et al. The design and synthesis of pyrazine amide ligands suitable for the ‘tiles’ approach to molecular weaving with octahedral metal ions. Inorg. Chem. Commun. 11, 1–4 (2008).
Wadhwa, N. R., Hughes, N. C., Hachem, J. A. & Mezei, G. Metal-templated synthesis of intertwined, functionalized strands as precursors to molecularly woven materials. RSC Adv. 6, 11430–11440 (2016).
Wang, Z. et al. Molecular weaving via surface-templated epitaxy of crystalline coordination networks. Nat. Commun. 8, 14442 (2017).
Lewandowska, U. et al. A triaxial supramolecular weave. Nat. Chem. 9, 1068–1072 (2017).
Acknowledgements
We thank J. E. Beves and J.-F. Ayme (now University of New South Wales and BASF SE, respectively) for early ligand designs for this target topology; the Engineering and Physical Sciences Research Council (EPSRC; EP/P027067/1), the European Research Council (ERC; Advanced Grant no. 786630), and East China Normal University for funding; the EPSRC National Mass Spectrometry Service Centre for high-resolution mass spectrometry; the Diamond Light Source for synchrotron beam time on I19 (XR029); networking contributions from the COST Action CA17139, EUTOPIA and Alberto Valero for the video of the rotating 74 knot structure. D.A.L. is a Royal Society Research Professor.
Author information
Authors and Affiliations
Contributions
J.J.D., S.D.P.F., J.-F.L. and S.L.W. carried out the synthesis and characterization studies. G.F.S.W. solved the crystal structure. D.A.L. directed the research. All the authors contributed to the analysis of the results and the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Experimental procedures and characterization data. Supplementary discussion, Figs. 1–40, Tables 1–3, spectra 1–21 and references 1–21.
Supplementary Video 1
Movie of endless 74 knot based on the X-ray crystal structure of the 3 × 3 grid with Merck Mechanics Force Field (MMFF)-modelled cyclized end groups.
Supplementary Data
3 × 3 grid crystallographic data.
Rights and permissions
About this article
Cite this article
Leigh, D.A., Danon, J.J., Fielden, S.D.P. et al. A molecular endless (74) knot. Nat. Chem. 13, 117–122 (2021). https://doi.org/10.1038/s41557-020-00594-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-00594-x
This article is cited by
-
Mechanical scission of a knotted polymer
Nature Chemistry (2024)
-
Cross-catenation between position-isomeric metallacages
Nature Communications (2024)
-
Synthesis of nanoparticles based on wavy interlocked macrocycles template
Applied Physics A (2024)
-
The sharp structural switch of covalent cages mediated by subtle variation of directing groups
Nature Communications (2023)
-
Using spatial and logical-mathematical intelligence for information wayfinding
Nature Chemistry (2023)