Abstract
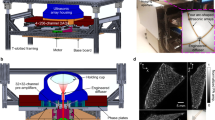
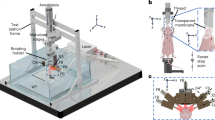
Imaging of small animals has played an indispensable role in preclinical research by providing high-dimensional physiological, pathological and phenotypic insights with clinical relevance. Yet, pure optical imaging suffers from either shallow penetration (up to ~1–2 mm) or a poor depth-to-resolution ratio (~3), and non-optical techniques for whole-body imaging of small animals lack either spatiotemporal resolution or functional contrast. Here, we demonstrate that stand-alone single-impulse panoramic photoacoustic computed tomography (SIP-PACT) mitigates these limitations by combining high spatiotemporal resolution (125 μm in-plane resolution, 50 μs per frame data acquisition and 50 Hz frame rate), deep penetration (48 mm cross-sectional width in vivo), anatomical, dynamical and functional contrasts, and full-view fidelity. Using SIP-PACT, we imaged in vivo whole-body dynamics of small animals in real time and obtained clear sub-organ anatomical and functional details. We tracked unlabelled circulating melanoma cells and imaged the vasculature and functional connectivity of whole rat brains. SIP-PACT holds great potential for both preclinical imaging and clinical translation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baker, M. Whole-animal imaging: the whole picture. Nature 463, 977–980 (2010).
Zanzonico, P. in Small Animal Imaging: Basics and Practical Guide (eds Kiessling, F. & Pichler, J. B. ) 3–16 (Springer, 2011).
Ntziachristos, V. Going deeper than microscopy: the optical imaging frontier in biology. Nat. Methods 7, 603–614 (2010).
Jun, X. & Wang, L. V. Small-animal whole-body photoacoustic tomography: a review. IEEE Trans. Biomed. Eng. 61, 1380–1389 (2014).
Wu, D. & Zhang, J. In vivo mapping of macroscopic neuronal projections in the mouse hippocampus using high-resolution diffusion MRI. Neuroimage 125, 84–93 (2016).
Alomair, O. I., Brereton, I. M., Smith, M. T., Galloway, G. J. & Kurniawan, N. D. In vivo high angular resolution diffusion-weighted imaging of mouse brain at 16.4 Tesla. PLoS ONE 10, e0130133 (2015).
Schambach, S. J., Bag, S., Schilling, L., Groden, C. & Brockmann, M. A. Application of micro-CT in small animal imaging. Methods 50, 2–13 (2010).
Brenner, D. J. & Hall, E. J. Computed tomography—an increasing source of radiation exposure. N. Engl. J. Med. 357, 2277–2284 (2007).
Greco, A. et al. Ultrasound biomicroscopy in small animal research: applications in molecular and preclinical imaging. J. Biomed. Biotechnol. 2012, 519238 (2012).
Kim, T. et al. White-light diffraction tomography of unlabelled live cells. Nat. Photon. 8, 256–263 (2014).
Horton, N. G. et al. In vivo three-photon microscopy of subcortical structures within an intact mouse brain. Nat. Photon. 7, 205–209 (2013).
Zhang, F. et al. Multimodal fast optical interrogation of neural circuitry. Nature 446, 633–639 (2007).
Darne, C., Lu, Y. & Sevick-Muraca, E. M. Small animal fluorescence and bioluminescence tomography: a review of approaches, algorithms and technology update. Phys. Med. Biol. 59, R1–R64 (2014).
Razansky, D. et al. Multispectral opto-acoustic tomography of deep-seated fluorescent proteins in vivo . Nat. Photon. 3, 412–417 (2009).
Wang, L. H. V. & Hu, S. Photoacoustic tomography: in vivo imaging from organelles to organs. Science 335, 1458–1462 (2012).
Yao, J. et al. High-speed label-free functional photoacoustic microscopy of mouse brain in action. Nat. Methods 12, 407–410 (2015).
Jathoul, A. P. et al. Deep in vivo photoacoustic imaging of mammalian tissues using a tyrosinase-based genetic reporter. Nat. Photon. 9, 239–246 (2015).
Yao, J. et al. Multiscale photoacoustic tomography using reversibly switchable bacterial phytochrome as a near-infrared photochromic probe. Nat. Methods 13, 67–73 (2016).
Brecht, H.-P. et al. Whole-body three-dimensional optoacoustic tomography system for small animals. J. Biomed. Opt. 14, 064007 (2009).
Taruttis, A., Morscher, S., Burton, N. C., Razansky, D. & Ntziachristos, V. Fast multispectral optoacoustic tomography (MSOT) for dynamic imaging of pharmacokinetics and biodistribution in multiple organs. PLoS ONE 7, e30491 (2012).
Merčep, E., Burton, N. C., Claussen, J. & Razansky, D. Whole-body live mouse imaging by hybrid reflection-mode ultrasound and optoacoustic tomography. Opt. Lett. 40, 4643–4646 (2015).
Razansky, D., Buehler, A. & Ntziachristos, V. Volumetric real-time multispectral optoacoustic tomography of biomarkers. Nat. Protoc. 6, 1121–1129 (2011).
Luis Dean-Ben, X. & Razansky, D. Adding fifth dimension to optoacoustic imaging: volumetric time-resolved spectrally enriched tomography. Light Sci. Appl. 3, e137 (2014).
Tang, J., Coleman, J. E., Dai, X. & Jiang, H. Wearable 3-D photoacoustic tomography for functional brain imaging in behaving rats. Sci. Rep. 6, 25470 (2016).
Yuan, X. & Wang, L. V. Effects of acoustic heterogeneity in breast thermoacoustic tomography. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 50, 1134–1146 (2003).
Cohn, J. N. et al. Noninvasive pulse-wave analysis for the early detection of vascular-disease. Hypertension 26, 503–508 (1995).
Kis, E. et al. Pulse wave velocity in end-stage renal disease: influence of age and body dimensions. Pediatr. Res. 63, 95–98 (2008).
Stein, E. W., Maslov, K. & Wang, L. V. Noninvasive, in vivo imaging of blood-oxygenation dynamics within the mouse brain using photoacoustic microscopy. J. Biomed. Opt. 14, 020502 (2009).
Xia, J. et al. Calibration-free quantification of absolute oxygen saturation based on the dynamics of photoacoustic signals. Opt. Lett. 38, 2800–2803 (2013).
Raichle, M. E. Behind the scenes of functional brain imaging: a historical and physiological perspective. Proc. Natl Acad. Sci. USA 95, 765–772 (1998).
Ogawa, S., Lee, T.-M., Kay, A. R. & Tank, D. W. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc. Natl Acad. Sci. USA 87, 9868–9872 (1990).
Roberts, M. B. V. in Biology: A Functional Approach 4th edn, 243–249 (Thomas Nelson and Sons, 1986).
Joiner, J. T. in NOAA Diving Manual: Diving For Science And Technology 4th edn, 1–36 (Best Publishing Company, 2001).
Karimova, A. & Pinsky, J. D. The endothelial response to oxygen deprivation: biology and clinical implications. Intensive Care Med. 27, 19–31 (2001).
Piantadosi, C. A. in The Biology of Human Survival: Life and Death in Extreme Environments 129–139 (Oxford Univ. Press, 2003).
Miura, G. Cancer tumor imaging: catch me if you can. Nat. Chem. Biol. 10, 485–485 (2014).
Nagrath, S. et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450, 1235–1239 (2007).
Kienast, Y. et al. Real-time imaging reveals the single steps of brain metastasis formation. Nat. Med. 16, 116–122 (2010).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 66, 7–30 (2016).
Srinivasan, V. J. et al. Quantitative cerebral blood flow with optical coherence tomography. Opt. Express 18, 2477–2494 (2010).
Cox, S. B., Woolsey, T. A. & Rovainen, C. M. Localized dynamic changes in cortical blood flow with whisker stimulation corresponds to matched vascular and neuronal architecture of rat barrels. J. Cereb. Blood Flow Metab. 13, 899–913 (1993).
Jonckers, E., Van Audekerke, J., De Visscher, G., Van der Linden, A. & Verhoye, M. Functional connectivity fMRI of the rodent brain: comparison of functional connectivity networks in rat and mouse. PLoS ONE 6, e18876 (2011).
Osmanski, B.-F., Pezet, S., Ricobaraza, A., Lenkei, Z. & Tanter, M. Functional ultrasound imaging of intrinsic connectivity in the living rat brain with high spatiotemporal resolution. Nat. Commun. 5, 5023 (2014).
Dean-Ben, X. L. et al. Functional optoacoustic neuro-tomography for scalable whole-brain monitoring of calcium indicators. Light Sci. Appl. 5, e16201 (2016).
Gottschalk, S., Fehm, T. F., Deán-Ben, X. L. & Razansky, D. Noninvasive real-time visualization of multiple cerebral hemodynamic parameters in whole mouse brains using five-dimensional optoacoustic tomography. J. Cereb. Blood Flow Metab. 35, 531–535 (2015).
Schackert, G., Price, J. E., Bucana, C. D. & Fidler, I. J. Unique patterns of brain metastasis produced by different human carcinomas in athymic nude mice. Int. J. Cancer 44, 892–897 (1989).
Anastasio, M. A. et al. Half-time image reconstruction in thermoacoustic tomography. IEEE Trans. Med. Imaging 24, 199–210 (2005).
Xu, M. & Wang, L. V. Universal back-projection algorithm for photoacoustic computed tomography. Phys. Rev. E 71, 016706 (2005).
Treeby, B. E. & Cox, B. T. K-Wave: MATLAB toolbox for the simulation and reconstruction of photoacoustic wave fields. J. Biomed. Opt. 15, 021314 (2010).
Xia, J., Huang, C., Maslov, K., Anastasio, M. A. & Wang, L. V. Enhancement of photoacoustic tomography by ultrasonic computed tomography based on optical excitation of elements of a full-ring transducer array. Opt. Lett. 38, 3140–3143 (2013).
Frangi, A. F., Niessen, W. J., Vincken, K. L. & Viergever, M. A. in International Conference on Medical Image Computing and Computer-Assisted Intervention (eds Wells, W. M., Colchester, A. & Delp, S. ) 130–137 (Springer, 1998).
Farnebäck, G. in Image Analysis (eds Bigun, J. & Gustavsson, T. ) 363–370 (Springer, 2003).
White, B. R. et al. Imaging of functional connectivity in the mouse brain. PLoS ONE 6, e16322 (2011).
Acknowledgements
We thank Y. He, C. Li, Y. Li and J. Xia for their technical support, and J. Ballard for close reading of the manuscript. This work was sponsored by the United States National Institutes of Health (NIH) grants DP1 EB016986 (NIH Director’s Pioneer Award), R01 CA186567 (NIH Director’s Transformative Research Award), U01 NS090579 (BRAIN Initiative), U01 NS099717 (BRAIN Initiative), R01 EB016963 and S10 RR026922.
Author information
Authors and Affiliations
Contributions
L.Li and L.V.W. conceived and designed the study. L.Li and L.Z. constructed the hardware system. L.Li, L.Z. and C.M. developed the software system and the reconstruction algorithm. L.W. and J.S. constructed the control program. K.M. and W.C. designed the preamplifiers. L.Li, C.M. and L.Lin performed the experiments. R.Z. cultured the B16 cells. L.Li, L.Z., C.M. and J.Y. analysed the data. L.V.W. supervised the study. All authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
L.V.W. and K.M. have a financial interest in Microphotoacoustics, Inc.; however, Microphotoacoustics, Inc. did not support this work. The other authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures, tables, references and video captions. (PDF 17806 kb)
Supplementary Video 1
In vivo label-free photoacoustic computed tomography of mouse internal organs. (MP4 47735 kb)
Supplementary Video 2
In vivo label-free photoacoustic computed tomography of a mouse's whole-body anatomy at a cross-section of the upper thoracic cavity. (MP4 18496 kb)
Supplementary Video 3
In vivo label-free photoacoustic computed tomography of mouse's whole-body anatomy at a cross-section of the lower thoracic cavity. (MP4 14849 kb)
Supplementary Video 4
In vivo label-free photoacoustic computed tomography of a mouse's whole-body anatomy at a cross-section of the liver. (MP4 5966 kb)
Supplementary Video 5
In vivo label-free photoacoustic computed tomography of a mouse's whole-body anatomy at a cross-section of the upper abdominal cavity. (MP4 6368 kb)
Supplementary Video 6
In vivo label-free photoacoustic computed tomography of a mouse's whole-body anatomy at a cross-section of the lower abdominal cavity. (MP4 13612 kb)
Supplementary Video 7
In vivo cross-sectional images of the mouse liver reconstructed by increasing angular coverage. (MP4 237 kb)
Supplementary Video 8
Pulse-wave-induced cross-sectional-area changes of two vertical arteries over time. (MP4 1761 kb)
Supplementary Video 9
In vivo label-free photoacoustic computed tomography of the mouse brain in response to an oxygen challenge. (MP4 3395 kb)
Supplementary Video 10
Oxygenation response of the lower abdominal cavity of a mouse during whole-body oxygen challenge. (MP4 7848 kb)
Supplementary Video 11
Label-free tracking of circulating melanoma tumour cells in the mouse brain in vivo. (MP4 8069 kb)
Supplementary Video 12
In vivo monitoring of dye perfusion in the mouse brain. (MP4 955 kb)
Supplementary Video 13
In vivo label-free photoacoustic computed tomography of rat whole-body anatomy at a cross-section of the lower abdominal cavity. (MP4 3008 kb)
Rights and permissions
About this article
Cite this article
Li, L., Zhu, L., Ma, C. et al. Single-impulse panoramic photoacoustic computed tomography of small-animal whole-body dynamics at high spatiotemporal resolution. Nat Biomed Eng 1, 0071 (2017). https://doi.org/10.1038/s41551-017-0071
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-017-0071
This article is cited by
-
Functional photoacoustic imaging: from nano- and micro- to macro-scale
Nano Convergence (2023)
-
Ultrafast longitudinal imaging of haemodynamics via single-shot volumetric photoacoustic tomography with a single-element detector
Nature Biomedical Engineering (2023)
-
Parallel interrogation of the chalcogenide-based micro-ring sensor array for photoacoustic tomography
Nature Communications (2023)
-
Non-invasive photoacoustic computed tomography of rat heart anatomy and function
Light: Science & Applications (2023)
-
Photoacoustic vector tomography for deep haemodynamic imaging
Nature Biomedical Engineering (2023)